A steam power cycle with regeneration as shown below on the $T-s$ diagram employs a single open feedwater heater for efficiency improvement. The fluid mix with each other in an open feedwater heater. The turbine is isentropic and the input (bleed) to the feedwater heater from the turbine is at state $2$ as shown in the figure. Process $3-4$ occurs in the condenser. The pump work is negligible. The input to the boiler is at state $5$. The following information is available from the steam tables:
$\begin{array}{|l|l|l|l|l|l|l|} \hline \text{State} & 1 & 2 & 3 & 4 & 5 & 6 \\ \hline \text{Enthalpy (kJ/kg)} & 3350 & 2800 & 2300 & 175 & 700 & 1000 \\ \hline \end{array}$
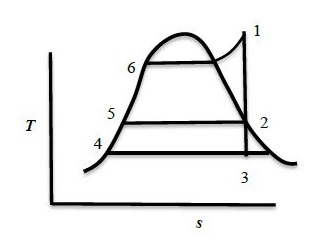
The mass flow rate of steam bled from the turbine as a percentage of the total mass flow rate at the inlet to the turbine at state $1$ is _______